Mod GRF (1-29) 5mg (CJC-1295 no DAC)

Mod GRF (1-29) 5mg (CJC-1295 no DAC)
each and save 4 %
each and save 6 %
each and save 10 %
FREE Shipping for orders over $200
Free (1) 30 ml Bacteriostatic Water
with qualified orders over $500 USD.
(excludes capsule products, cosmetic peptides, promo codes and shipping)
Modified GRF (CJC-1295 No DAC) is a truncated peptide analogue of growth hormone releasing hormone (GHRH). First developed in the 1980s, research studies with modGRF have shown it to improve muscle repair and growth, accelerate wound healing, strengthen bones, increase fat burning, and improve metabolism. It may also have beneficial effects on blood sugar regulation and the immune system.
Product Usage: This PRODUCT IS INTENDED AS A RESEARCH CHEMICAL ONLY. This designation allows the use of research chemicals strictly for in vitro testing and laboratory experimentation only. All product information available on this website is for educational purposes only. Bodily introduction of any kind into humans or animals is strictly forbidden by law. This product should only be handled by licensed, qualified professionals. This product is not a drug, food, or cosmetic and may not be misbranded, misused or mislabled as a drug, food or cosmetic.
What is Modified GRF (1-29)?
Modified GRF (1-29) is a peptide analogue of growth-hormone-releasing hormone (GHRH). Modified GRF was first developed in the 1980s when it was discovered that the first 29 amino acids of GHRH retained all of the properties of the full 44-amino-acid protein. The original protein, called GRF (1-29), is the smallest fragment of GHRH that retains all of the properties of GHRH. Another truncated synthetic form of GHRH called Sermorelin or GRF 1-29 has 29 amino acids; Modified GRF (1-29) is further modifeid in that it has four substituted aminos in its chain that serve the purposes of preventing degradation and oxidation in manufacture and transport as well as in vivo, while also increasing binding affinity to the GHRH receptors.
Though GRF had full effect, the duration of action was limited due to a very short half-life (less than 10 minutes). As a result, researchers modified GRF (1-29) to give it a longer half-life and thus greater therapeutic potential. Modified GRF (1-29) is occasionally referred to as mod GRF or tetrasubstituted GRF (1-29). The latter name makes it clear that mod GRF differs from GRF (1-29) as a result of four amino acids being changed. Modified GRF (1-29) is identical to CJC-1295 without DAC.
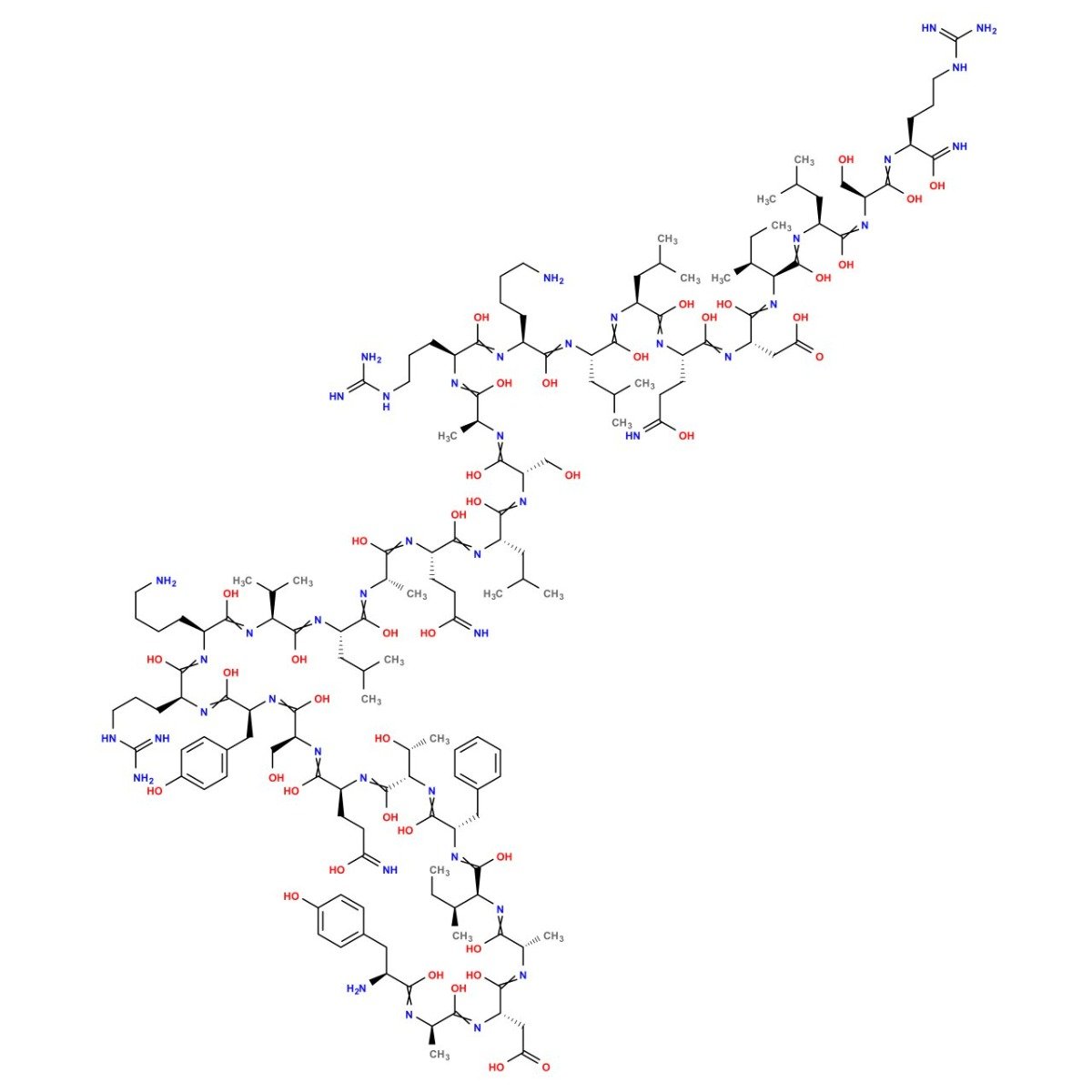
Modified GRF (1-29) Structure
Sequence: Tyr-D-Ala-Asp-Ala-Ile-Phe-Thr-Gln-Ser-Tyr-Arg-Lys-Val-Leu-Ala-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Leu-Ser-Arg
Sermorelin Sequence: Tyr-Ala-Asp-Ala-Ile-Phe-Thr-Asn-Ser-Tyr-Arg-Lys-Val-Leu-Gly-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Met-Ser-Arg
Molecular Formula: C152H252N44O42
Molecular Weight: 3367.954 g/mol
PubChem CID: 56841945
CAS Number: 863288-34-0
Modified GRF (1-29) Research
Modified GRF has the same effects as GHRH. These effects include muscle growth (hypertrophy), accelerated wound healing, improved bone growth, enhanced fat burning, and accelerated metabolism. Modified GRF also has positive effects on the immune system and blood sugar regulation.
1. Mod GRF and the Intestine
Early research suggested that GH had positive effects on Crohn’s disease, short bowel syndrome, and other inflammatory bowel conditions. Unfortunately, the benefits of GH in this setting are far outweighed by the side effects of long-term exogenous administration. This prompted researchers to look at whether GHRH analogues might have similar effects without the same serious side effects. Research in monkeys indicates that mod GRF interacts with vasoactive intestinal peptide (VIP) receptors and actually improves bowel motility. The peptide appears to interact with VIPC1, which is found on the smooth muscle of the gastrointestinal, reproductive, and urinary systems[1], [2]. Improved bowel motility is of substantial importance in inflammatory bowel diseases associated with constipation. These conditions lead to a great deal of morbidity and are currently difficult to treat due to lack of drug options.
2. Mod GRF and Heart Function
Research in rodent models suggests that modified GRF and other GHRH derivatives may improve ethe ability of the heart to pump blood following heart attack[3]. This could be of profound use in improving quality of life and reducing death over the long-term in individuals diagnosed with heart failure. The range of disease this finding could be applied to include pulmonary hypertension, heart failure, heart attack, and any number of congenital heart conditions.
3. The Interaction Between Thyroid Hormone and Growth Hormone
Problems with the thyroid gland are often associated with concomitant problems in GH release. To probe this interaction, scientists stimulated individuals suffering from hypothyroidism with modified GRF. They found that individuals given thyroid replacement hormone had stronger reactions to GRF, proving a link between thyroid hormone and GH [4]. This is just one of a number of ways that peptides like modified GRF can help scientists to better understand human physiology.
Modified GRF exhibits moderate side effects, low oral and excellent subcutaneous bioavailability in mice. Per kg dosage in mice does not scale to humans. Modified GRF for sale at Peptide Sciences is limited to educational and scientific research only, not for human consumption. Only buy Modified GRF if you are a licensed researcher.
Article Author
The above literature was researched, edited and organized by Dr. Logan, M.D. Dr. Logan holds a doctorate degree from Case Western Reserve University School of Medicine and a B.S. in molecular biology.
Scientific Journal Author
Dr. Jan Izdebski has been listed as a noteworthy Chemist, researcher by Marquis Who's Who. He attained a Master of Science from University Warsaw, Poland, 1959 and is a Doctor of Philosophy, University Warsaw, Poland, 1965, along with a Doctor of Science, University Warsaw, Poland, 1979. He studied how GRF 1-29 is more resistant to enzymatic degradation and how that affects its half-life.
Dr. Jan Izdebski is being referenced as one of the leading scientists involved in the research and development of GRF 1-29. In no way is this doctor/scientist endorsing or advocating the purchase, sale, or use of this product for any reason. There is no affiliation or relationship, implied or otherwise, between Peptide Sciences and this doctor. The purpose of citing the doctor is to acknowledge, recognize, and credit the exhaustive research and development efforts conducted by the scientists studying this peptide. Dr. Jan Izdebski is listed in [5] under the referenced citations.
Referenced Citations
- T. Ito et al., “GI side-effects of a possible therapeutic GRF analogue in monkeys are likely due to VIP receptor agonist activity,” Peptides, vol. 22, no. 7, pp. 1139–1151, Jul. 2001.
- M. Waelbroeck, P. Robberecht, D. H. Coy, J.-C. Camus, P. D. Neef, and J. Christophe, “Interaction of Growth Hormone-Releasing Factor (GRF) and 14 GRF Analogs with Vasoactive Intestinal Peptide (VIP) Receptors of Rat Pancreas. Discovery of (N-Ac-Tyr1,D-Phe2)-GRF(l-29)-NH2 as a VIP Antagonist,” Endocrinology, vol. 116, no. 6, pp. 2643–2649, Jun. 1985.
- A. V. Schally, X. Zhang, R. Cai, J. M. Hare, R. Granata, and M. Bartoli, “Actions and potential therapeutic applications of growth hormone-releasing hormone agonists,” Endocrinology.
- R. Valcavi et al., “Growth Hormone Responses to Grf 1–29 in Patients with Primary Hypothyroidism Before and During Replacement Therapy with Thyroxine,” Clin. Endocrinol. (Oxf.), vol. 24, no. 6, pp. 693–698, 1986.
- Izdebski, J. , Witkowska, E. , Kunce, D. , Orłowska, A. , Baranowska, B. and Wolińska‐Witort, E. (2004), Potent Trypsin‐resistant hGH‐RH Analogues. J. Peptide Sci., 10: 524-529.
ALL ARTICLES AND PRODUCT INFORMATION PROVIDED ON THIS WEBSITE ARE FOR INFORMATONAL AND EDUCATIONAL PURPOSES ONLY.
The products offered on this website are furnished for in-vitro studies only. In-vitro studies (Latin: in glass) are performed outside of the body. These products are not medicines or drugs and have not been approved by the FDA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law.